ILD / IPF

ILD Disease Process
Interstitial lung diseases (ILDs) encompass a large group of chronic lung conditions that affect the respiratory bronchioles, alveolar walls, and alveolar spaces. These diseases are characterized by progressive scarring (interstitial lung abnormaliites, fibrosis) of lung tissue which can lead to severe respiratory dysfunction.
VIDA’s comprehensive portfolio of quantitative imaging biomarkers for ILD & Idiopathic Pulmonary Fibrosis (IPF) aim to precisely measure physiological changes associated with these diseases, including tissue remodeling (e.g., fibrosis), vascular remodeling, airway remodeling and functional impairment. VIDA’s imaging biomarkers complement traditional non-imaging endpoint measures (e.g., spirometry).

Source: L Plantier, et al. “Physiology of the Lung in Idiopathic Pulmonary Fibrosis.” ERS Review 27, no. 147 (March 31, 2018).
VIDA’s Comprehensive Portfolio of ILD Imaging Biomarkers
Given the wide spectrum of Interstitial lung diseases (ILDs) and their complex pathophysiology, it is often useful for drug developers to evaluate a multifaceted set of imaging biomarkers. VIDA offers 30+ imaging biomarkers that are relevant to ILD/IPF. To learn about this full suite of ILD biomarkers, view our white paper.
Fibrosis Quantification
Fibrosis quantification, based on CT imaging, has grown to be the most common quantitative imaging biomarker in ILD therapeutic clinical trials. Approximately 50% of active, industry-funded, interventional ILD/IPF trials now include fibrosis quantification as a primary or secondary endpoint.
DTA Fibrosis Score
Data-driven Texture Analysis (DTA) Fibrosis Score, licensed from National Jewish Health, quantifies the % of lung volume (and mass) classified as fibrotic. Fibrosis is identified based on hallmark radiologic features, including reticulation, honeycombing, and traction bronchiectasis.
- DTA Fibrosis Score is exceptionally well validated, with an established minimal clinically important difference (MCID) among IPF cohorts of 3.4% (source)
- DTA Fibrosis Score is robust and repeatable across a broad range of ILD populations, with intraclass correlation coefficients of 0.78-0.91. (source)

Xenon MRI for ILD
In parnership with Polarean, VIDA offers innovative Xenon 129 MRI biomarkers. These biomarkers uniquely measure functional impact and therapeutic impact for ILDs.
| Ventilation Defect % (VDP%) | Interstitial Membrane Conductance (% High, % Low) |
|---|---|
| A quantitative analysis of xenon 129 gas distribution in the lungs. It can be used to evaluate and longitudinally monitor therapeutic response within weeks of the start date and at any frequency interval. It depicts areas where 129Xe is and is not distributing into the alveolar space.
|
This biomarker depicts the concentration of xenon 129 gas “trapped” in the lung membrane as seen in fibrotic lung disease, measuring the ratio of the membrane to the gas signal within each voxel. High or Low membrane conductance may suggest active fibrosis and/or inflammation in the lung parenchyma.
|
Mass-Based Tissue Scoring
VIDA has validated a novel mass-based ILD and fibrosis scoring methodology to complement traditional volume-based quantitative scores. This new mass-based tissue quantification, presented at ATS 2024 (source) , demonstrates an improvement in correlations between normal lung mass and non-imaging clinical assessments (FVC, FEV1 and more) compared to volume-based measures. In addition to tighter clinical correlations, the mass-based approach has a major advantage:
Mass is less susceptible to patients’ inspiration efforts. Inspired air volume is dependent on effort and has a significant impact on volume-based quantification, whereas lung mass is mostly consistent across the breath cycle.
In light of these results and advantages, VIDA now provides both volume and mass-based tissue quantification scores for ILD trials.

Novel Visualizations for ILDs
VIDA’s proprietary visualizations complement quantitative measures, providing compelling visual evidence of anatomical changes and therapeutic response. Topographic Multi-Planar Reformat (tMPR) and Subpleura View are two examples.
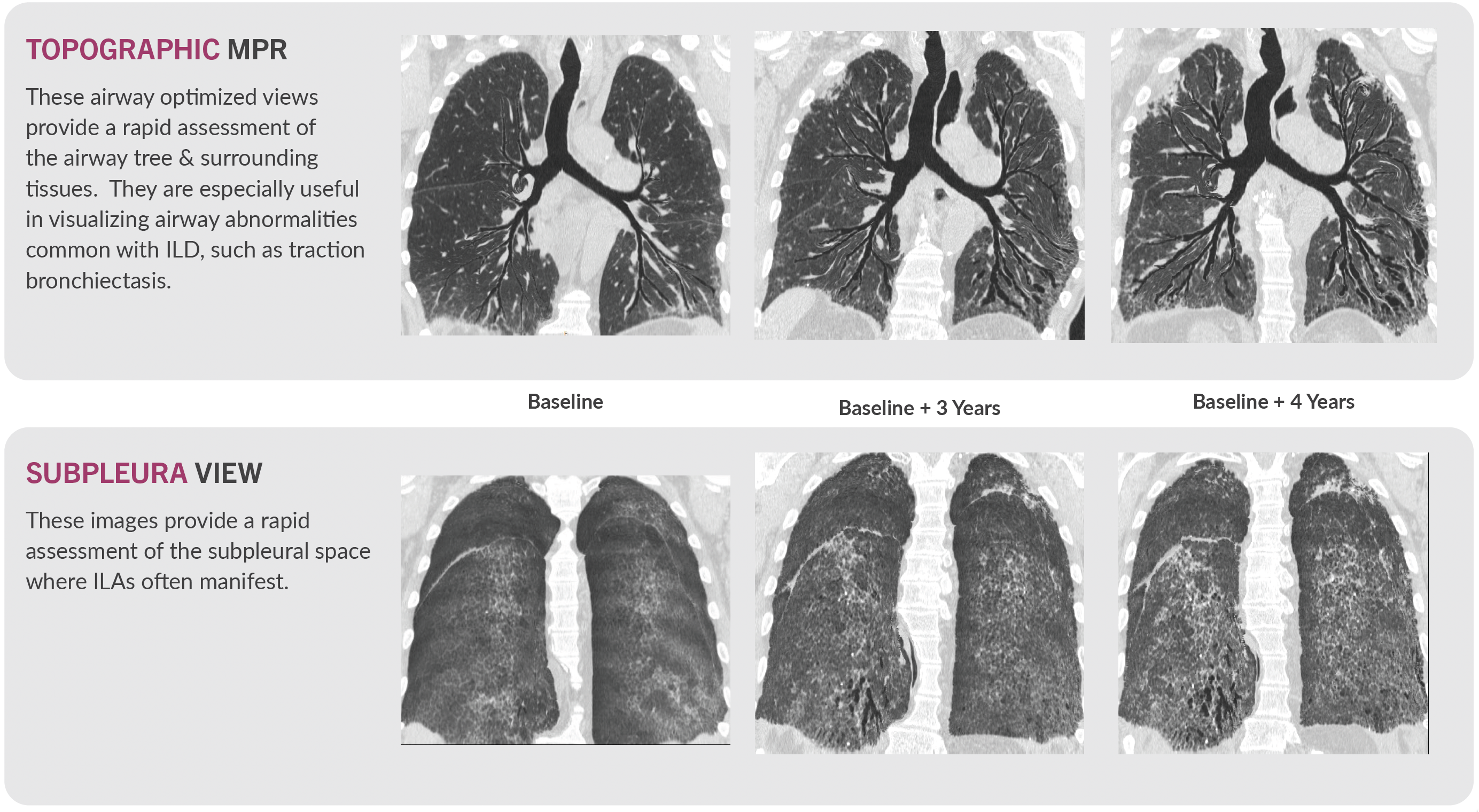
Data Quality
Biomarker Reliability
Reliability in quantitative CT (QCT) is determined by the consistency (or repeatability) of a measure. QCT measurement consistency (test/re-test) for 38 of VIDA’s digital imaging biomarkers was recently published in the American Journal of Respiratory and Critical Care Medicine. (4) While the cohort was a COPD cohort, many of the biomarkers also apply to ILD/IPF subjects. This important study showed high repeatability and reliability for QCT measures and revealed the importance of high-quality data and performing quality control (QC), which improved repeatability and reliability.

Modern Trial Imaging Management
Not all quantitative imaging data is equal.
To optimize data quality and streamline operational ease, VIDA provides a full range of trial imaging management services, built upon 20+ years of experience. These services are enabled by the VIDA Intelligence Platform, a modern AI-powered trial imaging system.
Since introduction of the innovative platform in early 2022:

*Among trials where imaging metrics are the primary endpoint.
VIDA is proud to provide the platform technology that powers OSIC Cloud, the world’s largest ILD imaging repository. OSIC Cloud is a product of the Open Source Imaging Consortium (OSIC), a cooperative effort between academia, industry and patient advocacy groups with a shared mission to accelerate progress against ILD/IPF.
More Resources
If you are interested to learn more about Quantitative Imaging Biomarkers for ILD/IPF, or our trial imaging services, we’d love to speak with you.
If addition, please enjoy the following programs and resources related to ILD/IPF:
ILD Studies Utilizing VIDA’s Precision Imaging










